AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Periodic table atomic number superscript8/12/2023  
The subscript tells the period number, the. Note how this simulation allows changing the atomic number and mass number. The ending gives the exact location of the atom on the periodic table. 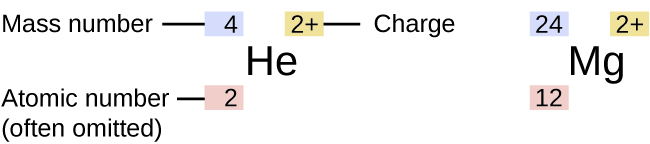
The simulation shows how the neutrons and protons must balance for the nucleus to be stable. This simulation builds atoms from protons, neutrons, and electrons and tests knowledge of the periodic table. t Elemental symbol Submit Hints My Answers Give Up Review Part When writing the symbol for an element, a superscript indicates the mass number and a subscript indicates the num atomic number. The University of Colorado has graciously allowed us to use the following PhET simulation. To find the number e of neutrons, the atomic number must be subtracted from the mass number. 'The Periodic Table, Its Story and Its Significance'. If you look at the periodic table, and you find the atomic number of 91, youll see that this is protactinium. 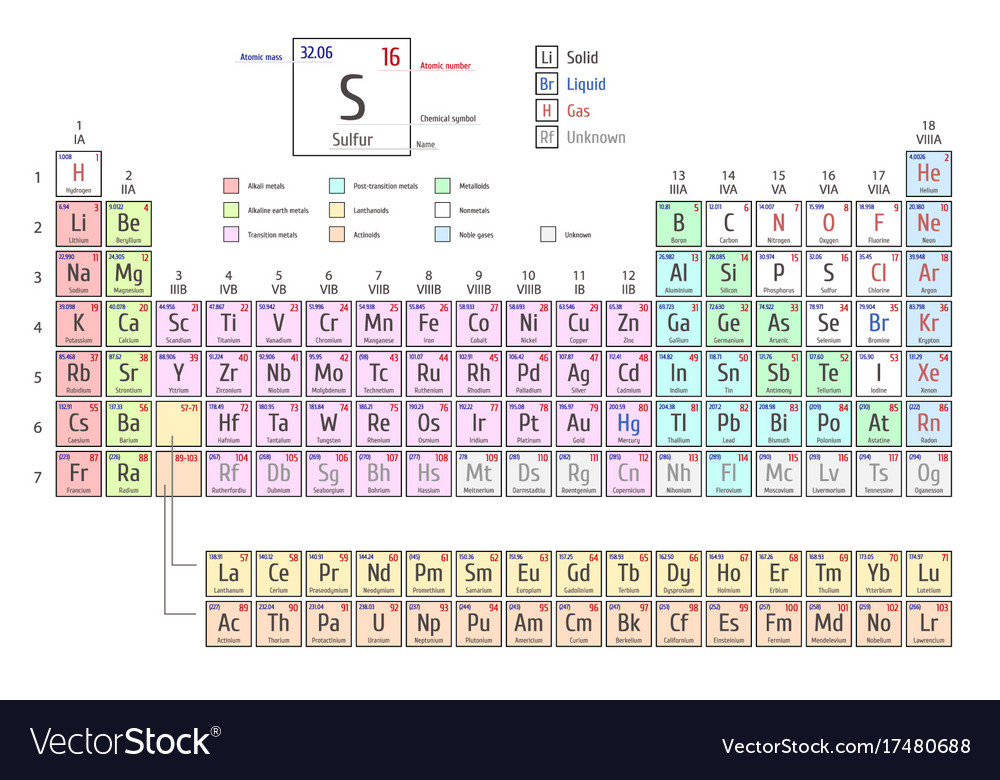
The atomic number is redundant to the chemical element, but is sometimes used to emphasize the change of numbers of nucleons in a nuclear reaction. The identity of the other product, just look it up here at our table, find atomic number of 90, and youll see thats thorium here. When it is included, the atomic number is put in a subscript before the name of the element and the superscript before the element (14 in this case) is the mass number:ĭifferent isotopes will have the same atomic number but different mass numbers an example would be: \ce. Attached subscripts or superscripts specifying a nuclide or molecule have the following meanings and positions. When writing out descriptions of a particular atom, sometimes the atomic number is included, and sometimes it's implied from the chemical name (if the chemical name is listed as nitrogen, the atomic number must be 7). ATOMIC NUMBER (Z): the number of protons in the nucleus of an atom whole numbers found on the periodic table identifies an element. Superscript numbers where appropriate but omit parentheses. For example, 6 is always carbon, 92 is always uranium, and 1 is always hydrogen.Ītomic number should not be confused with mass number, the total number of protons plus neutrons in a particular atom. configuration of the element with atomic number 8 in the periodic table. Each chemical element has a different number of protons, so the atomic number is a unique identifier for an element. The atomic number, written as Z, refers to the number of protons in the nucleus of an atom, and is used to organize the periodic table of elements. The periodic table is a chart that organizes elements by their atomic structure. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
